Metadata Report for BODC Series Reference Number 1047477
Metadata Summary
Problem Reports
Data Access Policy
Narrative Documents
Project Information
Data Activity or Cruise Information
Fixed Station Information
BODC Quality Flags
SeaDataNet Quality Flags
Metadata Summary
Data Description |
|||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||
Data Identifiers |
|||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||
Time Co-ordinates(UT) |
|||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||
Spatial Co-ordinates | |||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||
Parameters |
|||||||||||||||||||||||||||||||||||||||||||||||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||
Definition of BOTTFLAG | |||||||||||||||||||||||||||||||||||||||||||||||||
| BOTTFLAG | Definition |
|---|---|
| 0 | The sampling event occurred without any incident being reported to BODC. |
| 1 | The filter in an in-situ sampling pump physically ruptured during sample resulting in an unquantifiable loss of sampled material. |
| 2 | Analytical evidence (e.g. surface water salinity measured on a sample collected at depth) indicates that the water sample has been contaminated by water from depths other than the depths of sampling. |
| 3 | The feedback indicator on the deck unit reported that the bottle closure command had failed. General Oceanics deck units used on NERC vessels in the 80s and 90s were renowned for reporting misfires when the bottle had been closed. This flag is also suitable for when a trigger command is mistakenly sent to a bottle that has previously been fired. |
| 4 | During the sampling deployment the bottle was fired in an order other than incrementing rosette position. Indicative of the potential for errors in the assignment of bottle firing depth, especially with General Oceanics rosettes. |
| 5 | Water was reported to be escaping from the bottle as the rosette was being recovered. |
| 6 | The bottle seals were observed to be incorrectly seated and the bottle was only part full of water on recovery. |
| 7 | Either the bottle was found to contain no sample on recovery or there was no bottle fitted to the rosette position fired (but SBE35 record may exist). |
| 8 | There is reason to doubt the accuracy of the sampling depth associated with the sample. |
| 9 | The bottle air vent had not been closed prior to deployment giving rise to a risk of sample contamination through leakage. |
Definition of Rank |
|
|
Problem Reports
No Problem Report Found in the Database
Data Access Policy
Open Data
These data have no specific confidentiality restrictions for users. However, users must acknowledge data sources as it is not ethical to publish data without proper attribution. Any publication or other output resulting from usage of the data should include an acknowledgment.
If the Information Provider does not provide a specific attribution statement, or if you are using Information from several Information Providers and multiple attributions are not practical in your product or application, you may consider using the following:
"Contains public sector information licensed under the Open Government Licence v1.0."
Narrative Documents
Pall AcroPak 200, 500, 1000 and 1500 filter capsules with Supor membrane
AcroPak filter capsules are designed for sterile filtration. The capsule includes a Supor membrane (hydrophilic polyethersulfone, PES) which allows high flow rates and throughputs, and is ideal for solutions where low protein binding is required, although it is not recommended for use with some ketones. The capsule housing, vent plug, and support material are polypropylene, while the filling bell is polycarbonate. The unit is manufactured without the use of adhesives (sealing is achieved by thermal bonding) to minimize extractables, and each capsule is sterilised by gamma irradiation to eliminate potential contamination by EtO residuals before being packaged individually. A built-in prefilter extends filter life when particulate-laden solutions, such as serum-containing media, are processed.
AcroPak capsules are available in four versions, with different pore sizes, filtration areas and flow rates, as described below. The AcroPak 200 is suitable for low- to medium-volume filtration (up to 20 L), while the 500, 1000 and 1500 versions are suitable for high-volume filtration (up to 150 L).
Specifications
| Feature | AcroPak 200 | AcroPak 500 | AcroPak 1000 | AcroPak 1500 | |
|---|---|---|---|---|---|
| Pore size | 0.8/0.2 μm | 0.1/0.1, 0.2/0.2, 0.8/0.2 and 0.8/0.45 μm | 0.1/0.1, 0.2/0.2, 0.8/0.2 and 0.8/0.45 μm | 0.2/0.2 and 0.8/0.2 μm | |
| Effective filtration area | 200 cm2 | 500 cm2 | 1000 cm2 | 1500 cm2 | |
| Typical water flow rate (L min-1 at 0.1 bar) | 0.3 | 0.1/0.1 μm: 0.2 0.2/0.2 μm: 0.6 0.8/0.2 μm: 1.1 0.8/0.45 μm: 1.3 | 0.1/0.1 μm: 0.4 0.2/0.2 μm: 1.1 0.8/0.2 μm: 1.6 0.8/0.45 μm: 2.5 | 0.2/0.2 μm: 1.6 0.8/0.2 μm: 2.2 |
Further details can be found in the manufacturer's specification sheet.
Varian 810-MS and 820-MS inductively coupled plasma mass spectrometers (ICP-MS)
The Varian 810-MS and 820-MS systems are compact, floor-mounted inductively coupled plasma mass spectrometers (ICP-MS) featuring 90 ° reflecting ion optics systems for gigahertz sensitivity and low background and interferences. Each system comprises a sample introduction system, solid state 27 MHz RF generator, Turner Interlaced Coils and Discrete Dynode Electron Multiplier (DDEM) detector. The Varian 820-MS system also features a Collision Reaction Interface (CRI) providing fast, flexible, interference-free analysis using simple collision and reaction gases.
The 810-MS and 820-MS systems were manufactured by Varian, Inc., which was bought by Agilent Technologies in 2010.
Sample introduction
The External Sample Introduction Assembly (ESIA) comprises a spraychamber, nebuliser and peristaltic pump mounted outside the torch box to eliminate temperature-induced drift. The spraychamber is Peltier-cooled, with variable temperature control (from 15 °C to room temperature) for enhanced stability and reduced oxide ion interferences. The temperature is controlled by a PC and the spraychamber is insulated with inert polypropylene foam.
The glass concentric nebuliser allows flow rates of 100 μL min-1, while an inert PFA nebuliser and a range of low-flow, inert microconcentric nebulisers are optional. The peristaltic pump is also PC-controlled, with a variable speed from 0 to 50 rpm and three independently pressure adjustable channels for sample, drain, and internal standards or diluent.
RF generator
The 27.12 MHz solid-state, air-cooled, crystal locked RF generator is included in the main instrument housing. It has a range of 600-1600 W in 10 W increments. The user can customise the ignition sequence for different accessories and plasma types.
Plasma and plasma interface
Plasma parameters can be optimised according to user requirements (sensitivity, interferences, etc.). A PC controls the horizontal, vertical and sampling depth (Z position) of plasma for maximum sensitivity and minimum polyatomic interferences. For the cool plasma, the Turner Interlaced Coils minimise polyatomic interferences without the use of a mechanical torch shield.
The plasma interface is water-cooled for stability, including individual and independent cooling of the cones for faster warm-up, improved stability, and faster cool down. The 810-MS plasma interface comprises one set of C-series high performance Ni cones provided as standard, while platinum tipped C-series high performance cones are optional. One set of CRI cones is provided as standard in the 820-MS. Orifice diameters are 1.1 mm for the sampler and 0.5 mm for the skimmer.
Collision Reaction Interface
This is standard on the 820-MS and is available as an upgrade for the 810-MS. The CRI reduces interferences by injecting simple collision and reaction gases (hydrogen or helium) into the plasma as it passes through the orifice of the cones. Gas may be injected through the sampler cone, skimmer cone, or both and all CRI gas flows controlled by mass flow controllers. The CRI allows rapid switchover between gas on and gas off, or between different collision and reaction gases.
Ion optics
An ion mirror reflects the analyte ion beam through 90 ° while photons and neutrals pass to the vacuum system. The ion mirror creates parabolic electrostatic field to focus the analyte ions with optimum efficiency at the quadrupole entrance aperture, leading to gigahertz sensitivity (1000 Mc s-1 mg-1 L-1). All ion optics settings, including the ion mirror, are auto-optimised based on selected criteria such as signal and interferences. The quadrupole mass analyser is off-axis using curved stainless steel entrance rods to ensure low background by further eliminating excited neutrals before the quadrupole.
Vacuum system
All vacuum components located in the main housing of the instrument. There is a pneumatic vacuum isolation gate between the first and second vacuum stages, such that the gate automatically closes in the event of a power failure. The system includes two rotary (SD602 and SD302) and two V-301 turbomolecular pumps, with the first turbopump being positioned immediately behind the ion mirror and skimmer cone for maximum pumping efficiency and removal of unwanted neutrals and particles.
Quadrupole
This comprises precision-machined, stainless steel, round rods manufactured to micrometre tolerances and locked into ceramic mounts for a near-perfect hyperbolic field. Stainless steel construction permits determination of mercury without high memory. Curved entrance rods provide a double off-axis design and low background signals. All voltages are fully interlocked and under PC control. The system features a solid-state air-cooled power supply, a built-in, on board multi-channel scaler providing up to 40 channels per mass, range of 3 to 256 amu with "zero blast" protection and a resolution adjustable from 0.5 to 1.2 amu. The mass calibration stability is 0.05 amu per day, Quadrupole RF frequency is 3.0 MHz, scan speed is 2000 amu s-1, Quadrupole settling time is less than 400 μs, and minimum dwell time is 200 μs.
Detector
Both the 810-MS and the 821-MS include an all-digital ETP AF250 Discrete Dynode Electron Multiplier (DDEM) as standard. This provides nine decades of linear dynamic range in an all-pulse-counting detector, avoiding the need for analog-to-digital cross calibrations. Measuring dynodes are mounted off-axis for reduced background. The DDEM is fully controlled from a PC.
Performance
These details pertain to both the 810-MS and the 820-MS.
| Parameter | Hot plasma high sensitivity mode | Hot plasma normal sensitivity mode |
|---|---|---|
| Sensitivity (Mc s-1 mg-1 L-1) | 9Be >50 115In >1000 232Th >300 | 9Be >5 115In >50 232Th >50 |
| Precision (10 replicates, 20 minutes) | < 3% | < 3% |
| Long term stability (10 μg L-1 multi-element standard aspirated for 4 hours) | < 4% | |
| Oxides (CeO+/Ce+) | < 3% | < 1% (< 2% for CRI) |
| Doubly charged ions (Ba++/Ba+) | < 3% | < 3% |
| Background at 5 amu | < 10 | < 5 c s-1 |
Abundance sensitivity: 10-6 low, 10-7 high on 23Na
Isotope ratio precision: < 0.1%
CRI interference reduction: 1μg L-1 As readback in 1% HCl 1μg L-1 ± 0.1 μg L-1
Niskin Bottle
The Niskin bottle is a device used by oceanographers to collect subsurface seawater samples. It is a plastic bottle with caps and rubber seals at each end and is deployed with the caps held open, allowing free-flushing of the bottle as it moves through the water column.
Standard Niskin
The standard version of the bottle includes a plastic-coated metal spring or elastic cord running through the interior of the bottle that joins the two caps, and the caps are held open against the spring by plastic lanyards. When the bottle reaches the desired depth the lanyards are released by a pressure-actuated switch, command signal or messenger weight and the caps are forced shut and sealed, trapping the seawater sample.
Lever Action Niskin
The Lever Action Niskin Bottle differs from the standard version, in that the caps are held open during deployment by externally mounted stainless steel springs rather than an internal spring or cord. Lever Action Niskins are recommended for applications where a completely clear sample chamber is critical or for use in deep cold water.
Clean Sampling
A modified version of the standard Niskin bottle has been developed for clean sampling. This is teflon-coated and uses a latex cord to close the caps rather than a metal spring. The clean version of the Levered Action Niskin bottle is also teflon-coated and uses epoxy covered springs in place of the stainless steel springs. These bottles are specifically designed to minimise metal contamination when sampling trace metals.
Deployment
Bottles may be deployed singly clamped to a wire or in groups of up to 48 on a rosette. Standard bottles and Lever Action bottles have a capacity between 1.7 and 30 L. Reversing thermometers may be attached to a spring-loaded disk that rotates through 180° on bottle closure.
Trace Metal sampling on the Boundary voyage (Tan0609)
Originator's Protocol for Data Acquisition and Analysis
Samples were collected using acid-cleaned 5 litre, Teflon coated Niskin-X bottles attached to an autonomous rosette (Model 1018, General Oceanics Inc). The rosette was always stored under a plastic cover in a large plastic box. Prior to deployment the rosette was removed from the box, placed on a plastic pallet and the Nisken-X bottles fastened to the rosette. The bags covering Nisken bottle spigots were removed, the bottles cocked and then the large plastic rosette cover was removed. Upon retrieval, the rosette package was covered, the Nisken-X bottles removed from the rosette frame and racked in a trace-metal clean container with HEPA filtered air.
Sample filtration and storage
Seawater was filtered through acid-cleaned 0.2 µm capsule filters (Supor AcroPak 200, Pall) and acidified with quartz-distilled nitric acid. Samples bottles were stored at pH <1.8 for 10 months before dissolved trace metals analysis.
Trace metal determination
Dissolved trace metals were determined after dithiocarbamate solvent extraction (Bruland, et al., 1979; Danielsson, et al., 1978) by inductively coupled plasma mass spectrometry (ICP-MS) (820-MS, Varian).
Briefly, 100 g of seawater samples were buffered to a pH of 4.5 with purified ammonium acetate buffer. Purified ammonium pyrrolidinedithiocarbamate (APDC) and sodium diethyldithiocarbamate (DDC) were added to the samples. Samples were then extracted twice with purified chloroform by shaking for two 3 minute periods. Combined chloroform extracts were acidified with HNO3 and then diluted with deionised water.
Trace metal concentrations were determined by ICP-MS with hydrogen (60 ml/min-1) as the collision reaction interface gas (820-MS Varian, Australia) to reduce the interference of 40Ar16O on 56Fe. Isotopes used for reporting trace metals values were 56Fe, 59Co, 60Ni, 63Cu, 111Cd and 207Pb.
As a quality check, blanks and three SAFe reference samples (Johnson et al., 2007) were also analysed.
For full details please see Ellwood 2008.
References Cited
Bruland, K. W., R. P. Franks, G. A. Knauer, and J. H. Martin, 1979. Sampling and analytical methods for the determination of copper, cadmium, zinc and nickel at the nanogram per liter level in sea water. Anal. Chim. Acta, 105, 233-245.
Danielsson, L.-G., B. Magnusson, and S. Westerlund, 1978. An improved metal extraction procedure for the determination of trace metals in sea water by atomic absorption spectrometry with electrothermal atomization, Anal. Chim. Acta, 98, 47-57.
Ellwood, M. J., 2008. Wintertime trace metal (Zn, Cu, Ni, Cd, Pb and Co) and nutrient distributions in the Subantarctic Zone between 40-52°S; 155-160°E, Marine Chem., 112, 107-117.
Johnson, K. S., V. Elrod, S. Fitzwater, J. Plant, E. Boyle, B. Bergquist, K. Bruland, A. Aguilar-Islas, K. Buck, M. Lohan, et al., 2007. Developing Standards for Dissolved Iron in Seawater, EOS, 88, 131-132.
BODC Data Processing Procedures
Data were submitted to BODC in an Excel spreadsheet and archived under BODC's accession system. Sample metadata (CTD event, station number, date, sampling position and depth) were checked against information held in the cruise report and event logs. Where more than one event took place at a station, BODC added a suffix to the station number. Data were loaded into BODC's database using established BODC data banking procedures, time was converted from NZST (UTC/GMT +12 hours) to UTC.
A parameter mapping table is provided below;
| Originator's Parameter | Unit | Description | BODC Parameter Code | BODC Unit |
|---|---|---|---|---|
| DFe | nmol l-1 | Concentration of dissolved iron | CDFEICP1 | nmol l-1 |
| Zn | nmol l-1 | Concentration of dissolved zinc | CDZNICP1 | nmol l-1 |
| Cu | nmol l-1 | Concentration of dissolved copper | CDCUICP1 | nmol l-1 |
| Ni | nmol l-1 | Concentration of dissolved nickel | CDNIICP1 | nmol l-1 |
| Cd | pmol l-1 | Concentration of dissolved cadmium | CDCDICP1 | pmol l-1 |
| Co | pmol l-1 | Concentration of dissolved cobalt | CDCOICP1 | pmol l-1 |
| Pb | pmol l-1 | Concentration of dissolved lead | CDPBICP1 | pmol l-1 |
Data Quality Report
The data are of good quality, SAFe reference samples were run in triplicate. A number of data values were flagged by the originator who suspected them of being contaminated.
Project Information
GEOTRACES
Introduction
GEOTRACES is an international programme sponsored by SCOR which aims to improve our understanding of biogeochemical cycles and large-scale distribution of trace elements and their isotopes (TEIs) in the marine environment. The global field programme started in 2009 and will run for at least a decade. Before the official launch of GEOTRACES, fieldwork was carried out as part of the International Polar Year (IPY)(2007-2009) where 14 cruises were connected to GEOTRACES.
GEOTRACES is expected to become the largest programme to focus on the chemistry of the oceans and will improve our understanding of past, present and future distributions of TEIs and their relationships to important global processes.
This initiative was prompted by the increasing recognition that TEIs are playing a crucial role as regulators and recorders of important biogeochemical and physical processes that control the structure and productivity of marine ecosystems, the dispersion of contaminants in the marine environment, the level of greenhouse gases in the atmosphere, and global climate.
Scientific Objectives
GEOTRACES mission is: To identify processes and quantify fluxes that control the distribution of key trace elements and isotopes in the ocean, and to establish the sensitivity of these distributions to changing environmental conditions.
Three overriding goals support the GEOTRACES mission
- Determine ocean distributions of selected TEIs at all major ocean basins
- Evaluate the sources, sinks, and internal cycling of these TEIs and thereby characterize more completely their global biogeochemical cycles
- Provide a baseline distribution in the Polar Regions as reference for assessing past and future changes.
These goals will be pursued through complementary research strategies, including observations, experiments and modelling.
Fieldwork
The central component of GEOTRACES fieldwork will be a series of cruises spanning all Ocean basins see map below.
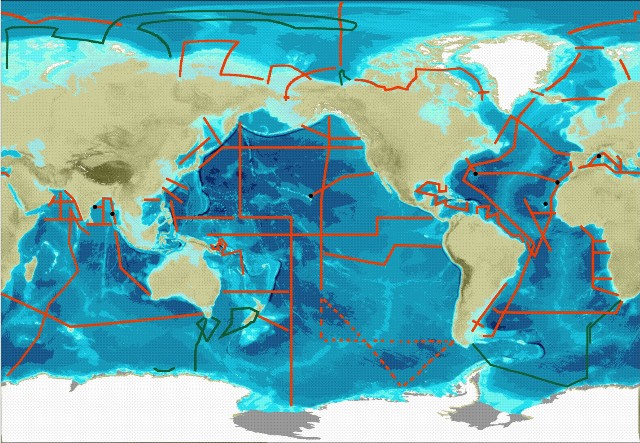
Three types of cruise are required to meet the goals set out by GEOTRACES. These are
- Section cruises - These will measure all the key parameters (see below) over the full depth of the water column. The sections were discussed and approved by the International GEOTRACES Scientific Steering Committee at the basin workshops.
- Process Studies - These will investigate a particular process relevant to the cycling of trace metal and isotopes. They must follow the "Criteria for Establishing GEOTRACES Process Studies" and be approved by the International GEOTRACES Scientific Steering Committee.
- Cruises collecting GEOTRACES compliant data - These will collect some trace element or isotope data. They must follow the GEOTRACES Intercalibration and Data Management protocols
IPY-GEOTRACES
The IPY-GEOTRACES programme comprised of 14 research cruises on ships from 7 nations; Australia, Canada, France, Germany, New Zealand, Japan and Russia. The cruises will not be classified in the same way as the full GEOTRACES programme since the intercalibration protocols and data management protocols had not been established before the start of the IPY. But IPY-GEOTRACES data will still be quality controlled by GDAC and in the majority of cases verified versus Intercalibration standards or protocols.
Key parameters
The key parameters as set out by the GEOTRACES science plan are as follows: Fe, Al, Zn, Mn, Cd, Cu; 15N, 13C; 230Th, 231Pa; Pb isotopes, Nd isotopes; stored sample, particles, aerosols.
Weblink:
http://www.bodc.ac.uk/geotraces/
http://www.geotraces.org/
Data Activity or Cruise Information
Data Activity
| Start Date (yyyy-mm-dd) | 2006-07-21 |
| End Date (yyyy-mm-dd) | 2006-07-21 |
| Organization Undertaking Activity | NIWA, Centre for Chemical and Physical Oceanography, University of Otago (now University of Otago, Research Centre for Oceanography) |
| Country of Organization | New Zealand |
| Originator's Data Activity Identifier | TAN0609_UCCTD_u4821c |
| Platform Category | lowered unmanned submersible |
BODC Sample Metadata Report for TAN0609_UCCTD_u4821c
| Sample reference number | Bottle rosette position | Bottle firing sequence number | Minimum pressure sampled (dbar) | Maximum pressure sampled (dbar) | Depth of sampling point (m) | Bottle type | Sample quality flag | Bottle reference | Comments |
|---|---|---|---|---|---|---|---|---|---|
| 227861 | 12 | 30.00 | 30.00 | 29.80 | 5-litre Niskin | No problem reported | |||
| 227862 | 11 | 60.00 | 60.00 | 59.50 | 5-litre Niskin | No problem reported | |||
| 227863 | 10 | 80.00 | 80.00 | 79.30 | 5-litre Niskin | No problem reported | |||
| 227864 | 9 | 100.00 | 100.00 | 99.20 | 5-litre Niskin | No problem reported | |||
| 227865 | 8 | 150.00 | 150.00 | 148.70 | 5-litre Niskin | No problem reported | |||
| 227866 | 7 | 200.00 | 200.00 | 198.30 | 5-litre Niskin | No problem reported | |||
| 227867 | 6 | 250.00 | 250.00 | 247.80 | 5-litre Niskin | No problem reported | |||
| 227868 | 5 | 300.00 | 300.00 | 297.30 | 5-litre Niskin | No problem reported | |||
| 227869 | 4 | 350.00 | 350.00 | 346.90 | 5-litre Niskin | No problem reported | |||
| 227870 | 3 | 400.00 | 400.00 | 396.40 | 5-litre Niskin | No problem reported | |||
| 227871 | 2 | 450.00 | 450.00 | 445.90 | 5-litre Niskin | No problem reported | |||
| 227872 | 1 | 500.00 | 500.00 | 495.30 | 5-litre Niskin | No problem reported |
Please note: the supplied parameters may not have been sampled from all the bottle firings described in the table above. Cross-match the Sample Reference Number above against the SAMPRFNM value in the data file to identify the relevant metadata.
Related Data Activity activities are detailed in Appendix 1
Cruise
| Cruise Name | TAN0609 (GIPY01) |
| Departure Date | 2006-07-12 |
| Arrival Date | 2006-08-05 |
| Principal Scientist(s) | Philip Boyd (NIWA, Centre for Chemical and Physical Oceanography, University of Otago) |
| Ship | Tangaroa |
Complete Cruise Metadata Report is available here
Fixed Station Information
No Fixed Station Information held for the Series
BODC Quality Control Flags
The following single character qualifying flags may be associated with one or more individual parameters with a data cycle:
| Flag | Description |
|---|---|
| Blank | Unqualified |
| < | Below detection limit |
| > | In excess of quoted value |
| A | Taxonomic flag for affinis (aff.) |
| B | Beginning of CTD Down/Up Cast |
| C | Taxonomic flag for confer (cf.) |
| D | Thermometric depth |
| E | End of CTD Down/Up Cast |
| G | Non-taxonomic biological characteristic uncertainty |
| H | Extrapolated value |
| I | Taxonomic flag for single species (sp.) |
| K | Improbable value - unknown quality control source |
| L | Improbable value - originator's quality control |
| M | Improbable value - BODC quality control |
| N | Null value |
| O | Improbable value - user quality control |
| P | Trace/calm |
| Q | Indeterminate |
| R | Replacement value |
| S | Estimated value |
| T | Interpolated value |
| U | Uncalibrated |
| W | Control value |
| X | Excessive difference |
SeaDataNet Quality Control Flags
The following single character qualifying flags may be associated with one or more individual parameters with a data cycle:
| Flag | Description |
|---|---|
| 0 | no quality control |
| 1 | good value |
| 2 | probably good value |
| 3 | probably bad value |
| 4 | bad value |
| 5 | changed value |
| 6 | value below detection |
| 7 | value in excess |
| 8 | interpolated value |
| 9 | missing value |
| A | value phenomenon uncertain |
| B | nominal value |
| Q | value below limit of quantification |
Appendix 1: TAN0609_UCCTD_u4821c
Related series for this Data Activity are presented in the table below. Further information can be found by following the appropriate links.
If you are interested in these series, please be aware we offer a multiple file download service. Should your credentials be insufficient for automatic download, the service also offers a referral to our Enquiries Officer who may be able to negotiate access.
| Series Identifier | Data Category | Start date/time | Start position | Cruise |
|---|---|---|---|---|
| 1047164 | Water sample data | 2006-07-21 01:42:00 | 45.213 S, 160.039 E | Tangaroa TAN0609 (GIPY01) |


